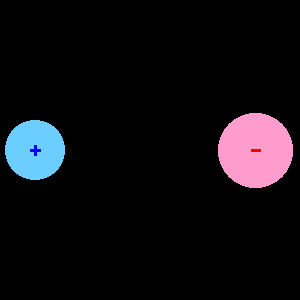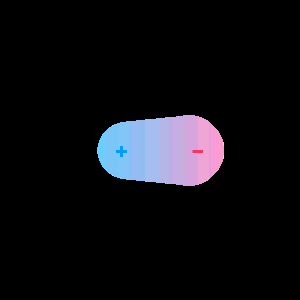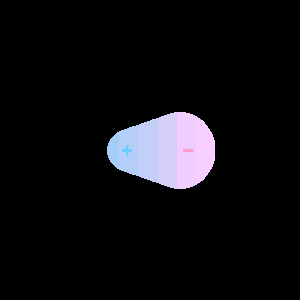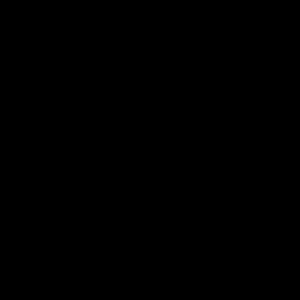
Versions 12 and higher have bromine-containing molecules. Versions 8 and higher have a few substituted benzenes with more than six heavy atoms. Six or fewer heavy atoms and twenty or fewer total atoms.See section I.B.1 for a periodic table view. Mostly atoms with atomic number less than than 36 (Krypton), except for most of the transition metals.Ions are indicated by placing + or - at the end of the formula (CH3+, BF4-, CO3-) The dipole moment is a useful concept when effects of microscopic charge separation are observable, but the actual distance between the charges is too small to be measured.A dipole moment is the distance between charges multiplied by the charge. A dipole is quantified by its dipole moment (). A comma delimited list of several species may be entered. A dipole is a separation of opposite electrical charges.Multiple specifications for an atom will be added.Parentheses may be used to group atoms. Querol and coworkers calculated changes in the quasi-electric dipole moment of a protein and used it as a simple criterion to predict stabilizing charge.If only one of a given atom is desired, you may omit. 
To specify the amounts of desired elements (e.g., C6H6). Enter a sequence of element symbols followed by numbers.The calculated dipole moments are reported as an unsigned total dipole and as three cartesiansįAQ Help Units Choose Units Dipole and Quadrupole to change dipole units. The electric dipole moment is a measure of the charge distribution in a molecule. For example, each of the two carbon-oxygen bonds in CO 2 has a dipole moment, but the CO 2 molecule has no dipole moment because the dipole moments of the two carbon-oxygen bonds are identical in magnitude and opposite in direction, resulting in a vector sum of zero. The form of this field is shown in Figure 5.34. If we rewrite it in terms of the dipole moment we get: E ( z) 1 4 0 p z 3. Dipole moment can arise in ionic bonds as well as in covalent bonds. Recall that we found the electric field of a dipole in Equation 5.7. A dipole moment occurs in any system in which there is a separation between the charges. You are here: Calculated > Electrostatics > Dipole > Dipole calculation typesĮlectric dipole moments calculated from atom chargesĬompared to dipoles from the wavefunction. A molecule may not have a dipole moment despite containing bonds that do. Figure 5.34 The net electric field is the vector sum of the field of the dipole plus the external field.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
